 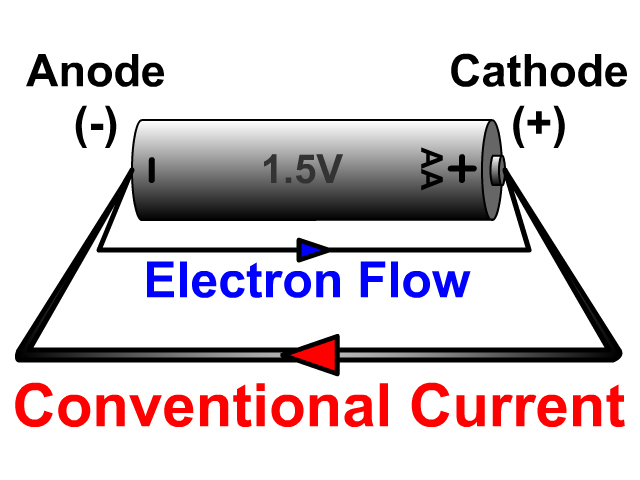
However, most batteries have some common components, although their material composition may vary. Therefore, each battery has a different composition. The chemical and material composition of batteries determines their size, format, and overall performance. Unlike a wet cell, a dry cell can operate in any orientation without spilling, as it contains no free liquid, making it suitable for portable equipment. A dry cell uses a paste electrolyte with only enough moisture to allow current to flow.Wet cells were a precursor to dry cells and are commonly used as a learning tool for electrochemistry. Other names are flooded cell since the liquid covers all internal parts, or vented cell since gases produced during operation can escape to the air. A wet cell battery has a liquid electrolyte.Many types of electrochemical cells have been produced, with varying chemical processes and designs, including galvanic cells, electrolytic cells, fuel cells, flow cells, and voltaic piles. The most common ones are lead, nickel, zinc, and lithium, each of them with different outputs and specific for some different purposes depending on the requirements. Batteries are designed so that the energetically favorable redox reaction can occur only when electrons move through the external part of the circuit.īatteries are made of an extensive range of materials resulting in different capabilities and behaviors in the functionality of the battery. 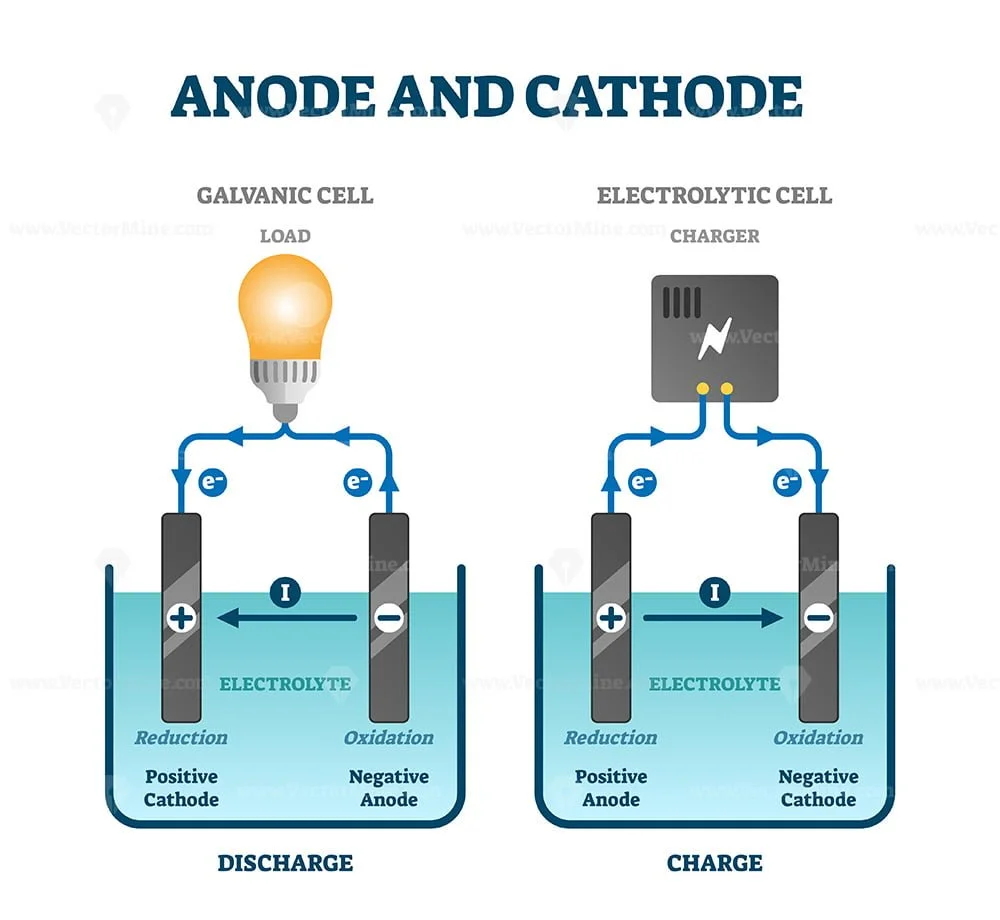
Even though many types of batteries exist with different combinations of materials, all of them use the same principle of the oxidation-reduction reaction. The fundamental principle in an electrochemical cell is spontaneous redox reactions in two electrodes separated by an electrolyte, which is a substance that is ionic conductive and electrically insulated.Ĭhemical energy can be stored, for example, in Zn or Li, which are high-energy metals because they are not stabilized by d-electron bonding, unlike transition metals. A typical battery consists of one or more voltaic cells. This then provides a source of electromotive force to enable currents to flow in electric and electronic circuits. It converts stored chemical energy into electrical energy through an electrochemical process. An electric battery is essentially a source of DC electrical energy. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
